 
Ěspartic acid - Asp - D (AsparDic acid - hard t sound like D).The importance of Gly and Pro in protein folding has been discussed by ( Krieger et al., 2005).Ĭharged amino acids (side chains often form salt bridges, in RED mnemonic rules to remember the one-letter code) : Sometimes Pro is called a helix breaker since it is often found at the end of α-helices. The reason for this is that its side chain makes a covalent bond with the main chain, which constrains the phi-angle of the polypeptide in this location (see the section of the Ramachandran plot). In contrast to Gly, which provides the polypeptide chain high flexibility, Pro provides rigidity by imposing certain torsion angles on the segment of the structure ( Morgan & Rubenstein, 2013). Proline, although considered hydrophobic, is also found at the surface, presumably due to its presence in turn and loop regions. 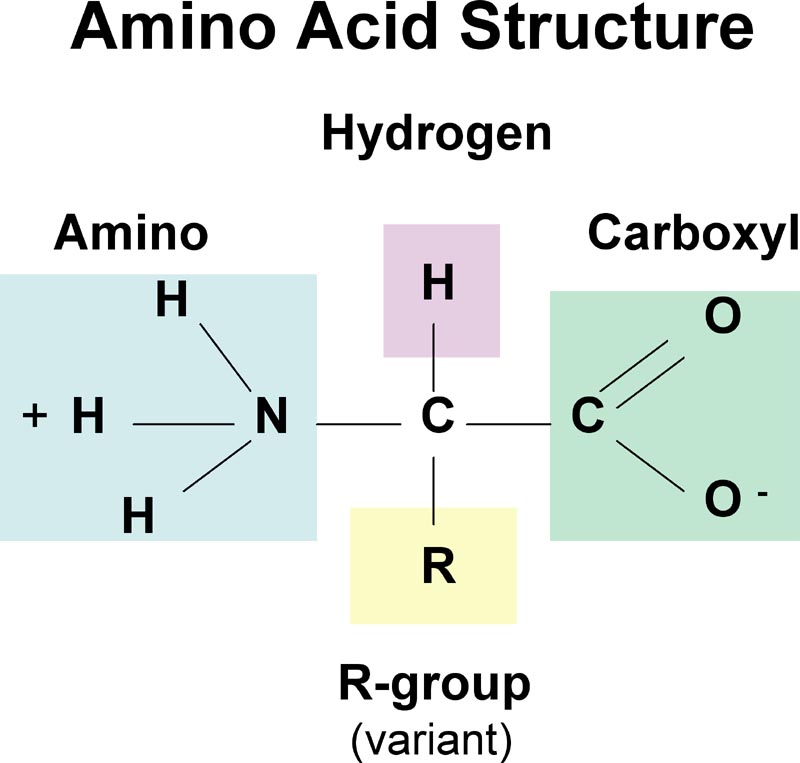
This flexibility is required in sharp polypeptide turns in loop structures. Glycine (Gly), one of the common amino acids, does not have a side chain and is often found at the surface of proteins within loop or coil regions (regions without defined secondary structure), providing high flexibility to the polypeptide chain. The image below showing the distribution of buried/exposed fractions of amino acids in protein molecules suggests that Cys is rather exposed. For example, according to some schemes, it is found in the interior due to its high reactivity (Fiser et al., 1996), while others consider it polar (REF). Also, in the case of Cys, some disagreement exists on its assignment to the hydrophobic group. Another essential function of Cys is metal binding, sometimes in enzyme active sites and sometimes in structure-stabilizing metal centers. In addition, Cys residues are involved in three-dimensional structure stabilization through the formation of disulfide (S-S) bridges, which sometimes connect different secondary structure elements or different subunits in a complex. The side chains within the core are tightly packed and participate in van der Waals interactions, which are essential for stabilizing the structure. These residues typically form the hydrophobic core of proteins, which is isolated from the polar solvent. The hydrophobic amino acids include alanine (Ala, A), valine (Val, V), leucine (Leu, L), isoleucine (Ile, I), proline (Pro, P), phenylalanine (Phe, F) and cysteine (Cys, C). They are also highly conserved within protein families, with Trp having the highest conservation rate. A characteristic feature of aromatic residues is that they are often found within the core of a protein structure, with their side chains packed against each other. For a discussion of OH−π and CH−O kinds of hydrogen bonds, see Scheiner et al., 2002. We should also note here that the side chains of histidine, and tyrosine, together with the hydrophobic phenylalanine and tryptophan, can also form weak hydrogen bonds of the types OH−π and CH−O, using electron clouds within their ring structures. In protein molecules, these residues are often found close to the interface between a protein and solvent. The aromatic amino acids tryptophan (Trp, W) and Tyr and the non-aromatic methionine (Met, M) are sometimes called amphipathic due to their ability to have both polar and nonpolar character. This ability makes histidine useful in enzyme active sites when the chemical reaction requires proton extraction. Within protein molecules, the pKa may be modulated by the environment so that the side chain may give away a proton and become neutral or accept a proton, becoming charged. When both groups are protonated, the side chain has a charge of +1. It has two –NH groups with a pKa value of around 6. Histidine (His, H), on the other hand, depending on the environment and pH, can be polar or carry a charge. Asparagine (Asn, N) and glutamine (Gln, Q) also belong to this group and may donate or accept a hydrogen bond. Tyrosine is also involved in metal binding in many enzymatic sites. Furthermore, this group can form a hydrogen bond with another polar group by donating or accepting a proton (a table showing donors and acceptors in polar and charged amino acid side chains can be found at the FoldIt site. For example, serine (Ser, S), threonine (Thr, T), and tyrosine (Tyr, Y) are polar since they carry a hydroxylic (-OH) group. 
When considering polarity, some amino acids are straightforward to define as polar, while in other cases, we may encounter disagreements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
